As potential coronavirus vaccines undergo development and testing, a shortage of dry ice has created uncertainty over how the vaccines will be transported.
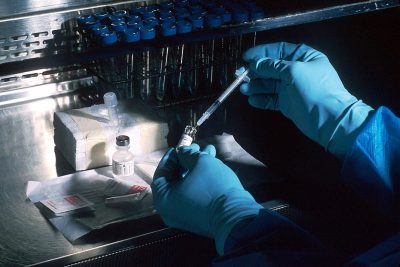
The vaccines are frozen at minus 70 degrees Celsius using dry ice.
Local ice suppliers have noticed shifts in supply and demand. Marc Savenor, owner of Acme Dry Ice in Cambridge, said he recognized a problem as early as March.
While demand for dry ice increased with vaccine research, Savenor said the supply temporarily fell due to production delays.
“By not driving cars and not making ethanol and not making energy, the supply of [carbon dioxide] was depleting,” Savenor said, “and [manufacturers] weren’t producing any [dry ice], because it’s a byproduct of all those things.”
Warmer months have brought cautious stability to the markets, Savenor said. Aside from seasonal customers who use dry ice for food, Savenor said new customers are interested in biotechnology and coronavirus research.
“Definitely with [COVID-19], it has picked up a lot,” Savenor said. “The demand has gone up, and we’ve been satisfying the demand.”
Tim Koerner, chief sales and service operator at American Carbonation, said he’s also concerned about the current market.
Koerner said a diminished supply of ethanol, a key component in producing carbon dioxide, halted dry ice production.
“Some of the ethanol plants have come back, but some of them haven’t,” Koerner said, “and there’s still definitely a big CO2 shortage.”
He added that supply cannot keep up with the current demand.
“These CO2 plants, they’re big projects when you build them,” Koerner said. “It’s not like you can just pop up a CO2 plant overnight. It takes big investment, big planning.”
While multiple pharmaceutical companies are developing their own unique mRNA-based vaccines, storage in sub-zero Celsius temperatures remains a common denominator among most companies, Mansoor Amiji, chairman of the Department of Pharmaceutical Sciences at Northeastern University, said.
“When you’re shipping long distances, obviously you need something that can sustain those lower temperatures for a longer duration,” Amiji said. “There, dry ice tends to be the best substitute.”
Beyond COVID-19 vaccine development, the applications of dry ice in a laboratory setting are broad, Amiji said. It can be useful in the safe transportation of flu vaccines, as well as in experiments that require rapid drops in temperature.
Moderna Therapeutics in Cambridge, however, is developing a COVID-19 vaccine that won’t require dry ice.
“mRNA is more stable, even in warmer temperatures, than protein,” Amiji said. “So there’s some hope for a vaccine like that.”






















































































































John Doe • Oct 1, 2020 at 6:29 pm
CO2 is there to make the dry ice. It’s the spike demand on the food supply industry that is consuming the ice supply. Does every American really need to have gourmet Omaha Steaks delivered to their door? That’s one of the biggest dry ice consumers in probably all of the world. Suppliers need to prioritize customers in these times. There are other resources for CO2 which comes in the form of a byproduct of ammonia. If we increase our farming, which utilizes ammonia, and figure out a way to do more farming in the winter months or ship ammonia to warmer climates, it in turn yields a higher demand for ammonia. More ammonia means more CO2. Dry ice shortage solved.